|
9/2/2023 0 Comments Complete ionic equation 
So this year represents my Net Ionic equation. Ion acquis gives me calcium sulfate solid. When we bring things down, will be s 04 to minus acquis, plus calcium. They also will not be part of the net Ionic. Ionic Also we have here to chlorides and to chlorides. So here, ammonium ion here matches up with ammonium ion here. Remember, spectator ions are the compounds that exists is both reactant and products at the same time. At this point, what we have is the complete ionic equation for the net. We're gonna cancel out the spectator ions from the complete Ionic equation in orderto isolate the Net Ionic equation. A quiz gives us The two gets distributed to each one of these ions here, so we're gonna have to ammonium ions. Plus, we have a two here to chloride ions. Plus, we're gonna have sulfate ion acquis plus one calcium ion. But remember this little to here, so there's actually two ammonium ions. So here remember, the coefficient gets distributed to each of these ions we're gonna have here one ammonium ion, acquis. So everyone breaks up into ions except for calcium sulfate, which is a solid. Now we're going to go to step five where we break up on Lee the acquis compounds into the respective ions. So here we have coefficients of one for the other compounds. The sulfate There's just one sulfate and one sulfate, one calcium and one calcium. Ammonium is here and to chorines here, So I have to put a two right here. Next we just have to balance out this molecular equation. Since we made a solid, a reaction has occurred. Since sulfate here is connected to calcium, it's gonna form a precipitate. Remember, CBS stands for calcium, barium or strontium. So this is a quiz, and then we have calcium sulfate based on the rules that we learned about calcium sulfate, remember, it's gonna form a precipitate if sulfate ion is connected to CBS. 
So remember, based on our side ability rules, anything connected to the ammonium ion will be soluble. At this point, we have to remember our saw You bility rules to see if we created Ah, solid liquid or gas. The numbers and the charges again are the same. So here, that will give me an H four c l plus, Then we're gonna have here calcium ion and sulfate ion. So when they connect together, remember when the numbers are the same, they just simply cancel out. Plus one charge is attracted to this minus one charge. Now, remember, we're gonna swap Ionic Partners because opposite charges attract this. We'll discuss that later on as we're going into the complete Ionic equation, and then calcium chloride breaks up into calcium ion and chloride ion. Don't worry about the fact that we have to Ammonium is here. 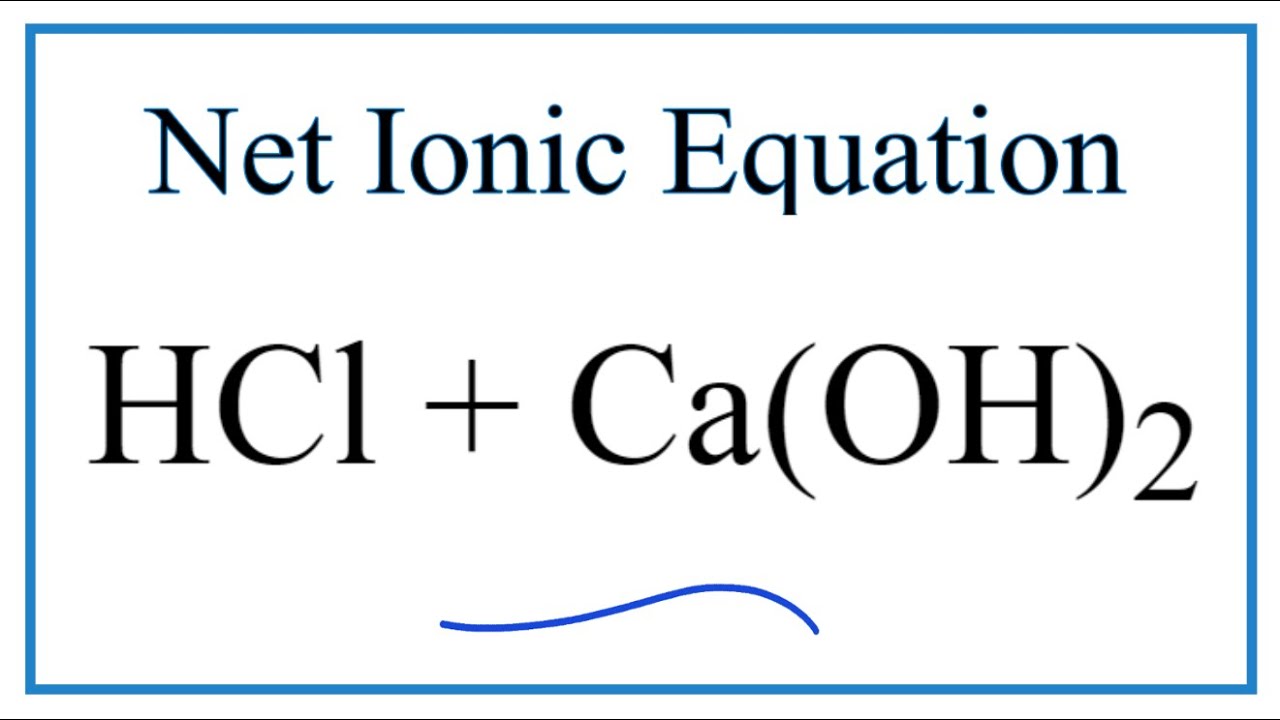
So ammonium sulfate breaks up into the ammonium ion and the sulfate ion. Now, remember, when it comes to these steps were first going to break up each of these compounds into their ionic forms. So for steps zero, we're just going to follow steps 1 to 4 that we've learned in the past to first give the molecular equation. 
So here we have ammonium sulfate react with calcium chloride. Remember to break up on Lee Equus compounds and remember to distribute the ions to each one of distribute number of coefficients to each one of those ions that you form.īased on the given reactant provide both the molecular equation and the complete ionic equation. So this year represents our complete ionic equation. So plus one so one not 61 calcium phosphate solid. So it stays together, does not break up in tow ions. So six lithium plus one ions plus six bromide ions. Remember, phosphate is a poly atomic ion produces six gets distributed to lithium bromide. So we know that's going to give us six lithium ions because it is two times three plus two phosphate ions. This too is gonna get distributed to hear and to hear. Plus, also, remember that when we have an eye on, it's an aqueous phase within a solution. Then we distribute the coefficient, so that's gonna give us three calcium ions and then this is three times to six bromide ions. When this breaks up into its ions, we know that we're gonna have calcium ion, a quiz plus bromide ion a quiz. Not what exactly does that mean? Well, here that means that this three is gonna get distributed to the number of calcium and the number of bro means so it gets distributed to see, um c a N b r. Also, remember that the coefficient gets distributed to each one of these compounds. If you are solid a liquid or gas, you will not break up in the complete ionic equation. All right, so remember, we can Onley break up acquis compounds so only these three compounds will break up. So here we have three moles of calcium bromide, acquis reacting with two moles of lithium phosphate, acquis producing six moles, a little lithium bromide as an acquis compound plus one mole of calcium phosphate. Question, it says convert the following molecular equation into a complete ionic equation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
